We Have Medical Device Treatment to Address ADHD Medication Shortages
A straightforward argument
Welcome to The Frontier Psychiatrists—a newsletter about health, health policy, and medical innovations; a psychiatrist writes it. Owen Scott Muir, M.D. is that M.D. and his training was completed in Child, Adolescent, and General Psychiatry.
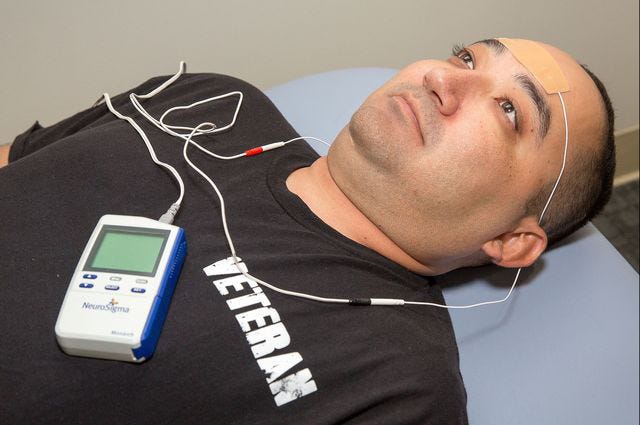
Today, we address the stimulant shortage with an actual solution—I’m describing a medical device that treats ADHD. This is a reader-supported newsletter, and I have no financial relationship with the company that makes it. I’m not an advisor, equity holder, or the like. I have no financial conflict of interest, but I’ll take one!1 I have a COI2 with you, dear readers, in that your subscriptions bias me towards more writing about topics like this. The device, made by Neurosigma, looks like this:
My practice, Fermata, specializes in noninvasive neuromodulation. In the context of ADHD, we prescribe the Monarch eTNS device. The Monarch device is FDA- approved3 for kids 7 through 12. The approval is as monotherapy—instead of medication—but it can be prescribed, like all FDA-approved treatments— off-label for adults with ADHD. My team at Acacia Mental Health also prescribe it—and we provide validated direct employer contracting!
It’s like a fancy TENS units you remember from 70s ads for nonsense abdominal fitness products. However, it’s way lower power—it creates an electrical current via sticky conductive pads on your forehead. You don’t turn it up much—just barely enough to feel a tingle.
The device pulses in a pattern which, via cranial nerve V division 1, resets your brain networks to be able to pay more attention. YES, I KNOW, IT IS SCIENCE! I am excited too. Feel free to come to see this authorial doctor about it.
How effective is Monarch eTNS?
In half of kids, it's twice as good as stimulant medications4.
If physicians prescribe this medical device, which is not a controlled substance, they may avoid stimulants in many cases.
More prescribing the Monarch eTNS device, pharmacists everywhere yell! It is possible to reduce hassle for ADHD treatment. Given stimulant shortages, the only real answer I can see is getting more effective treatments to more people—that don't require going through the DEA and supply chain constraints. This brain stimulator is the best treatment I've seen able to offer that at scale, and it’s available now.5. Move past stimulants? Onward to non-invasive brain stimulation!
Stimulants are a huge pain in the ass. Medical devices like Monarch are safer for ADHD. In the case of Monarch, it is “minimal risk!” One sleeps with a little pad on your head and the stimulator beside you. It is like a more comfortable CPAP device—for your attention.
What we prescribe first matters—because people stick with what works. Stimulant medications work. They also create a lot of extra work for physicians and stress for families! Every stimulant prescription—these days—means there will be a subsequent game of “find the pharmacy” to fill the script. Families are stalking pharmacies that legally can’t say what will be in stock, thanks to a settlement in the opioid lawsuits!
What does it cost?
The monarch device costs about $1000. The refills of the pads are the other expense— which is about $45/7 days. For kids 7-12, this can be covered with your insurance or a coupon for your insurance —if it's commercial. For adults, it's out of pocket.
For comparison, Mydayis (triphasic amphetamine) is around $400/month as its list price.
This is how we solve the ADHD treatment crisis—with better treatments!
I don't think we solve ADHD at scale by doing things that aren't scalable. At this point, there's a hard limit on stimulant medications, and there's a hard limit on the amount of frustration anybody is going to put up with to prescribe them.
Let’s prescribe the best treatment first—especially if it relieves hassle for health professionals and patients. And yes, I’d prescribe it for my kids too.
I enjoy a good conflict of interest disclosure, but I suspect conceptual and ideological disclosures matter a lot more when it comes to bias.
A conflict of interest
Yes, a de novo approval with studies behind it.
Efficacy and tolerability were first described in an 8-week, open-label, pilot study of eTNS treatment of 24 children diagnosed with ADHD aged 7-14 years1
Participants were assessed weekly with parent-and physician-completed measures of ADHD symptoms and executive functioning, treatment compliance, adverse events, and side effects.
After four weeks of nightly use, 64% of the study group were rated as “improved” or “improved very much” on the Clinical Global Impression–Improvement (CGI-I) scale.
After eight weeks, 71% had achieved that rating.
ADHD Rating Scale-IV (ADHD-RS-IV) scores showed a 47% decrease, compliance was 100%, and no child withdrew from the study due to adverse events
I'm also about to publish a paper on getting ADHD into remission—in 5 days— with transcranial magnetic stimulation. I was surprised about that too. I don't know how long that effect will be durable…


Thanks for sharing this information. I have been following with interest the development of non-invasive neurological stimulation for severe depression, PTSD, and ADHD. I’m also curious whether it may be of value with adults struggling with persistent anxiety related to childhood trauma.
I'm very excited to be prescribing the Monarch device and was compelled to do so by you. So, thank you. Also, the ones I have prescribed are on back order for over one month. Not exactly a solution to the stimulant shortage.
I'm concerned about the way that psychiatric is again being stigmatized and our patients are being treated like they don't have real issues. It is a real issue that kids who have difficulty effectively socializing with peers without treatment are going back to school without meds and maybe setting themselves up for a year of low self-esteem. Traffic accidents are real issues.
Stimulants are the safest and most effective medications we have in psychiatry. It feels a little gross to use this opportunity as a way to hype a product (that I can't actually get for my patients right now).