You Can Cure* Inpatient Psychiatry With this One Weird Open Comment Period
*The word "cure" is not intended to indicate the ability to treat any medical disease or condition. The word is used in jest as it relates to payment models only.
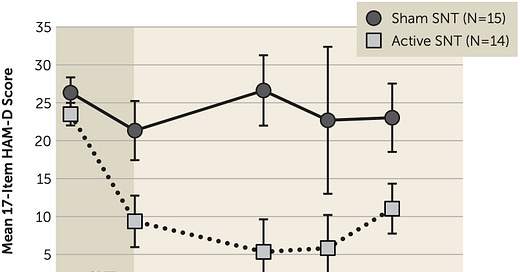
The TL: DR: Make a comment supporting SAINT depression treatment payment in hospitals here.
The marketers behind scams understand that we do not trust them. They build their arguments by taking mistrust into account.
When promising extraordinary weight loss with nonsense herbal supplements, they target our suspicion using terminology like “using this one weird trick.”
The assumption behind that phrase is that, for particularly credulous readers, who are the target audience for scam weight loss products, the concept that there may be just one thing they're missing is very appealing. Scammers have optimized their understanding of our mistrust. They acknowledge that the solutions is “weird” to assuage incredulity.
Most healthcare workers are justifiably even more mistrustful of extraordinary claims in the general public and are debatably less credulous.
If I were to tell you that inpatient psychiatry could be vastly better, and all you had to do was one thing, I imagine you would be justly sk…