The Future Will Be Neuromodulated: Medication-Free ADHD Treatment is FDA Cleared NOW
Great news for ADHD without meds: the Monarch eTNS Device.
The back-and-forth with the drug enforcement administration has been…robust. We had a public health emergency. And now we don't have one. We had a prescribing of controlled substances by telehealth. And now we may be don’t, but I will take credit for personally increasing the total number of comments submitted to regulators by 0.00052%. This has led to additional time for consideration. We have had shortages of controlled substances, and it's made it difficult for people with ADHD to get their medicine. With all due apology to “as seen on TV products” to open jars:
If only there were a better way!
At least for ADHD, we have one answer. Stimulant medications are remarkably effective for treating some attention deficit hyperactivity disorder symptoms of inattention and hyperactivity. Limited data exist to support the benefit in executive function. One psychotherapy works for executive dysfunction: organizational skills training (OST). (link to manual!). Other medicines are less effective and have different side effect profiles. If only there were another way?!?
I want to tell you there was a treatment for attention deficit hyperactivity disorder in youth. What if I told you that it is FDA-cleared right now? What if I were to tell you it was not a controlled substance?
What if it wasn't a drug at all?
Well, would you pay 29.99? I’m just kidding; this is Healthcare. It will be more, but less than My Taltz.
Let me tell you about transcutaneous neural stimulation!
Remember TENS units? They create an electrical current, it tingles on your skin, and if you turn it up enough, it makes muscles contract. It was introduced as a massage tool and exercise adjunct on TV for your abdominal area for decades. At much lower power, it stimulates sensory nerves and does not cause facial contractions.
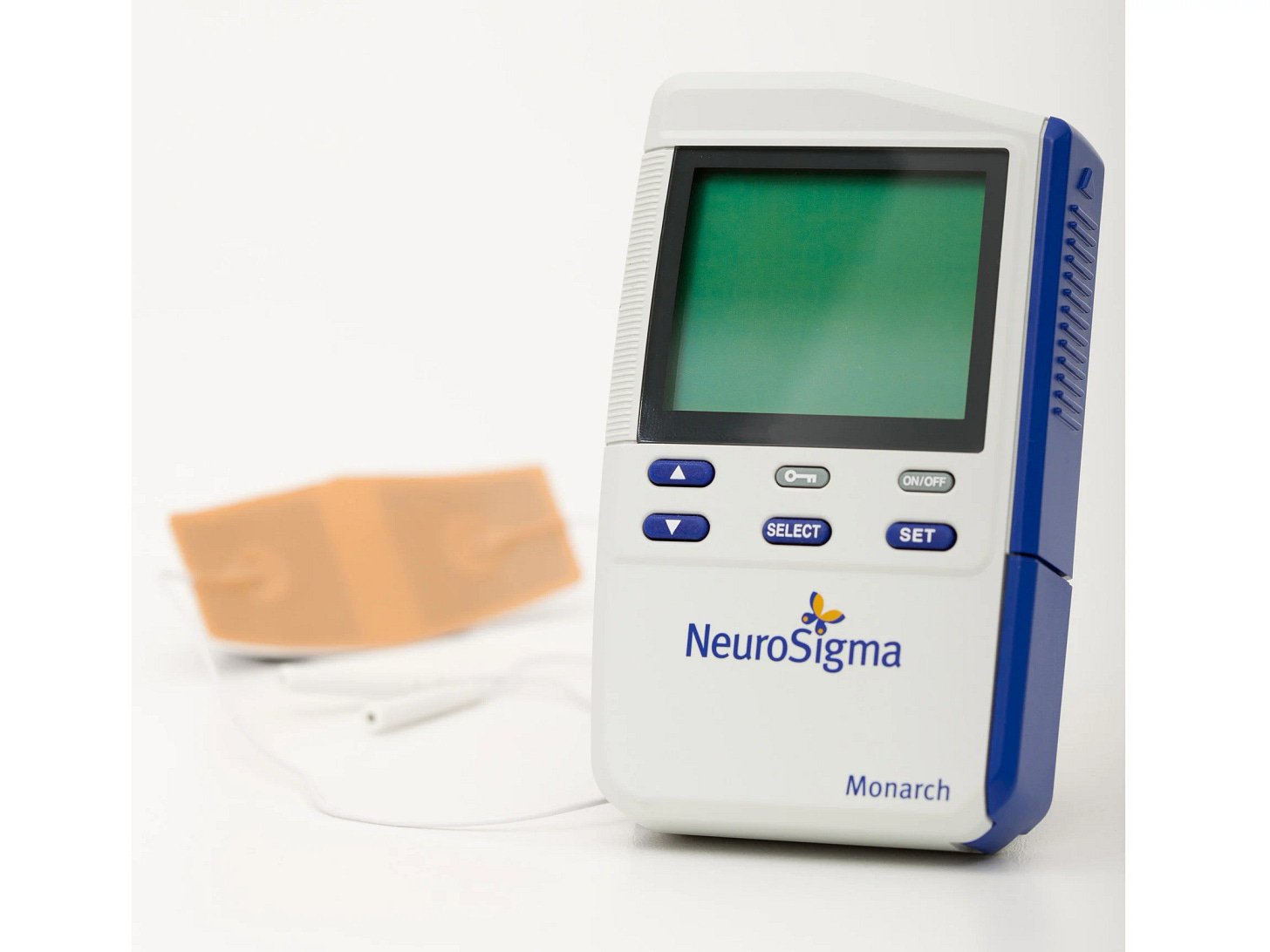
The Monarch eTNS Device!
The Monarch eTNS (external Trigeminal Nerve Stimulation) device is an innovative, non-invasive medical device that has been FDA-approved for the treatment of Attention Deficit Hyperactivity Disorder (ADHD) in children aged 7 to 12 years who are not currently on medication for the condition.
That is right—Monotherapy approval…instead of drugs. It’s by prescription only.
Developed by NeuroSigma, Inc., this technology aims to provide a safe and effective alternative to traditional pharmacological treatments for ADHD.
The Monarch eTNS system works by delivering mild electrical stimulation to the trigeminal nerve (Division V1), which is sensory innervation for the forehead. The device comprises a small, portable stimulator connected to a patch that sticks on your forehead. The electrical signals are transmitted through the patch, stimulating the trigeminal nerve. The device is worn in your sleep, and it’s an at-home treatment. I try mine tonight.
The device has an FDA clearance. The clinical trials have demonstrated the effectiveness of the Monarch eTNS device in reducing ADHD symptoms, such as inattention, hyperactivity, and impulsivity. It has minimal side effects that are generally mild and short-lived, including skin irritation and mild headaches.
The potency of the treatment’s description will allow me to use the Muir-Skee Low Emotionally Corrective Equation (M-SLo EQE). This allows us to convert an effect size of treatment into “difference in height” for easy and intuitive understanding.
The Monarch eTNS Device works exceptionally well in about half of kids. In the other half, it’s so-so. In the “super responders,” the effect size is 1.6. Stimulants are 0.8-0.9. This means if the device made you “a little bit taller,” using the M-SLo EQE, it would make you 4 inches taller. In the “average” case, it’s “only” 0.5 SMD effect size (about 1.25 inches). This is as potent as SSRIs in the first episode of depression.
This should be a first-line treatment. If it works well for any individual child, it works better than any medication for any condition in all of psychiatry. It’s not a drug. It has very limited side effects. It is a revolution in ADHD for kids and another example of the Neuro-modulated future of our field. Kids deserve access to a more effective and safe treatment first.
—Owen Scott Muir, M.D.
(Yes, we offer this treatment at my practice Fermata…link for inquiries—it is not yet cleared in Adults and thus can’t be advertised for off-label use).