Spravato
Our ongoing series on medications takes the jump into next-generation treatments
The Frontier Psychiatrists is a daily health-themed publication. I’ve been writing an extremely popular series for a very tiny niche audience on medications. This started as an attempt to explain what I could for my colleagues about the treatments I was no longer excited to use…I’ve jumped ship to lean into neuromodulation-first care at Radial!
Esketamine is the first FDA-approved “psychedelic-like” compound. The parent compound, Ketamine, is a dissociative anesthetic agent, about which I have senior-authored a review1, available in the American Journal of Therapeutics. I started working with Esketamine back in 2019, if memory serves, and have done research (as a Principal Investigator) related to safety and monitoring technology powered by AI.2
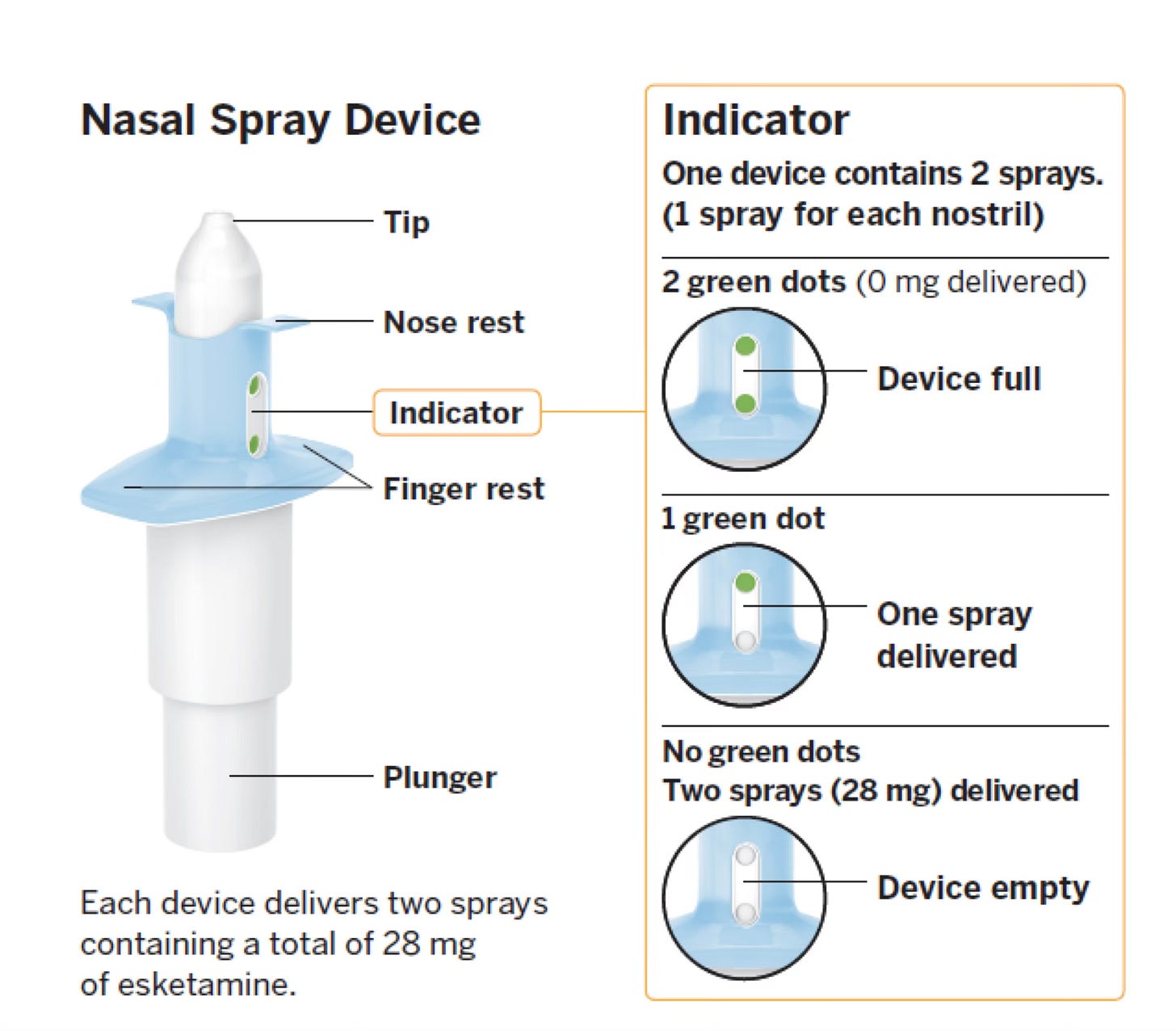
Now that the above study has been published, I’m free to write this article for you, dear readers, as a guide to this novel medication. The drug is available, unlike ketamine, only as a nasal spray at this time:
It has two FDA-Approved indications—through its breakthrough pathway—as of this writing, the first was treatment-resistant depression:
SPRAVATO (TM) is a non-competitive N-methyl D-aspartate (NMDA) receptor antagonist indicated, in conjunction with an oral antidepressant, for the treatment of treatment-resistant depression (TRD) in adults.
Defined as Adults with treatment-resistant depression (MDD patients who have had an inadequate response to 2 or more oral Antidepressants).
Its second was for Major Depressive Disorder with Suicidal Ideation (“MDSI”):
Depressive symptoms in adults with MDD with acute suicidal ideation or behavior (MDSI)
And, for reasons I will explain, it is only approved (yet) in adults who have concurrent oral antidepressant treatment.
It’s a drug, for people who have failed antidepressants, that has to be given, according to its label, only to people who are concurrently being given medicine that historically didn’t work.
Q.
I know.
“…”
I know. I know. Let me finish the Article.
“…”
🙏
When you are submitting a brand new treatment to the FDA as the “sponsor,” to treat a life-threatening condition, you can’t do anything …pardon the colloquialism, crazy. In this context, the word is used to mean reckless or unwise. We also do not allow new epilepsy treatments to enroll patients in randomized controlled trials that compare unproven new treatments alone versus placebo in someone with a serious and potentially life-threatening illness. People have complained to me, at times, of feeling like medical care was akin to being “experimented on”—this is exactly wrong, but not in the direction people imagine. In research, there are significant protections for human subjects! We have a whole certification process to be allowed near a human subject in a research trial. It is not ethical, once there are gold-standard treatments, to just try anything new in a serious medical condition, on average. At the very least, these studies require strict oversight. These oversight bodies are called institutional review boards. They are tasked with protecting people from renegade experiments. In the real world, after the experiments are done, patients, seen in clinical practice, have far fewer protections than those enrolled in “experiments.”
Thus, if you are Janssen, and you want to run a study to determine if your new compound is an effective treatment for suicidally depressed people, the study needs to include a standard-of-care treatment. To demonstrate that esketamine, on its own, is an effective treatment? That is another step.
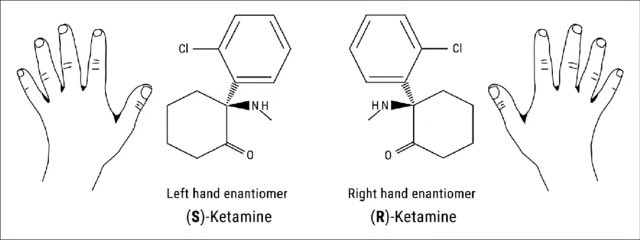
Before we go too much further—what is the difference between ketamine and esketamine anyway? Yes, Owen is going to explain organic chemistry, again.
Prior “orgo” primers (for all you pre-meds out there) include this.
When you do a chemical synthesis, there are some “substitution and elimination” reactions (Sn1) That lead to a mix of right and left-handed molecules. A mix! Ketamine is one such mixture—50% left-handed and 50% Right-handed. We refer to these as (R) and (S) “enantiomers” in chemistry. That (s) doesn’t have a vowel, so when we want a drug name to denote it’s just the “left-hand” version, we use “es” instead of just "S”. Thus…es-ketamine.
Behold!
Are you with me? Close enough, I am sure.
When a drug company, for reasons either cynical or scientific, or both, wants to take an old off-patent compound and make it a new compound, one method to do so it to purify the compound to only include the R or S enantiomer, and that counts as a new drug. Witness: Citalopram? > EScitalopram! Modafinil? ARmodafinil!
You get the idea. This is not entirely academic, however. If Esketamine can be a new drug, why not (r)-ketamine?3 Well, when Atai tried that, it didn’t work:
atai Life Sciences (NASDAQ: ATAI)… a clinical-stage biopharmaceutical company focused on mental health, announced that, while PCN-101 (R-ketamine) demonstrated signals of efficacy across all time points out to two weeks, Perception Neuroscience’s Phase 2a clinical trial did not meet its primary endpoint of a statistically significant change from baseline in participants’ MADRS (Montgomery-Åsberg Depression Rating Scale) score at 24 hours compared to placebo.
There are many reasons a trial will fail, and the arketamine story is unlikely to be over yet. But that is not what was demonstrated with the now FDA-approved EsKetamine.
Does Spravato work in treatment-resistant depression?
Very “yes.”
Results: Five trials with 774 patients were pooled. Adjunctive esketamine was significantly more effective than placebo for MADRS score change, response, and remission (N = 774, SMD = 0.36, P < .0001; response: risk ratio [RR] = 1.40, P < .0001; remission: RR = 1.45, P < .0001).4
That “smd” is standard mean difference—also known as an effect size! Thus, I can use the Muir-Skee Lo Emotionally Corrective Equation:
If esketamine were a medication for height, it would add 0.9 inches to your height.
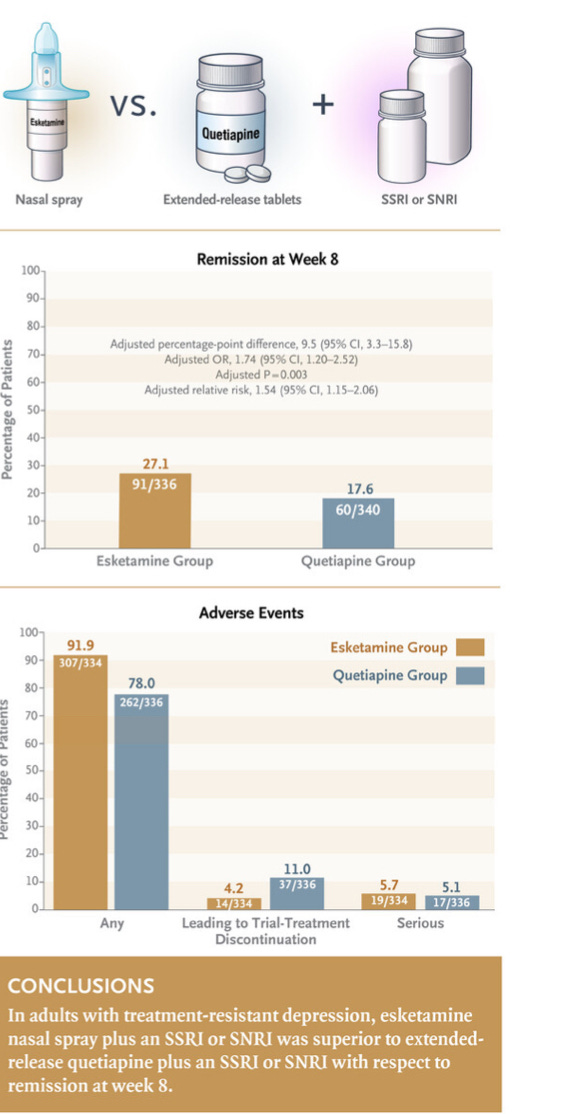
This is in a population where other treatments don’t work very often, which makes this more of a big deal than it might be otherwise. The New England Journal of Medicine published and open label comparison RCT in 2023 vs quetiapine. Esketamine was both safer and more effective5. NEJM is getting into the info graphic game, and it's good:
It could use a black cat or two, but they are new to this publication thing.
In the MDSI indication, there were two approval trials—ASPIRE I6and II.7 The finding that's worth noting is that although depression symptoms improved significantly, suicidality was not significantly different between placebo and active esketamine.
In ASPIRE I:
The difference between groups in the severity of suicidality was not statistically significant (median of treatment difference [95% CI]: 0.0 [-1.00 to 0.00]; 2-sided P = .107).
And in ASPIRE II:
Patients in both treatment groups experienced rapid reduction in Clinical Global Impression-Severity of Suicidality-revised score; the between-group difference was not statistically significant.
This is a surprising result, but it's why the label is for major depressive disorder with suicidal ideation—the population identified has depression get better as the claim—not reduction of suicidality. One could choose to be bummed about this, or you could recognize that the extremely potent nature of nonspecific factors in the treatment of suicidal individuals work. Feeling cared for, a research trial, if you're suicidal, is powerful in itself. It's so powerful that it's hard for any drug to demonstrate superiority over the power of human caring.
Esketamine is not cost effective
Spravato is a very expensive drug. There are two doses, 56 mg and 84 mg. The 84 mg dose—which almost everyone need—is about $886 a dose. The first month is 8 of these before we pay for the mandatory 2 hour monitoring period in a physician office that also requires ...payment. The upside is that insurance will usually pay for this and not the vastly less expensive Ketamine.
This has been evaluated for cost effectiveness…in QALYs (as addressed yesterday).
Over 5 years, esketamine was projected to increase time in remission from 25.3% to 31.1% of life-years, resulting in a gain of 0.07 QALYs. Esketamine increased societal costs by $16,617 and health care sector costs by $16,995. Base case ICERs were $237,111/QALY (societal) and $242,496/QALY (health care sector). Probabilistic sensitivity analysis showed a greater than 95% likelihood that esketamine’s ICER would be above $150,000/QALY. At a cost-effectiveness threshold of $150,000/QALY, esketamine’s value-based price was approximately $140/dose (versus a current price of $240/dose).8
How does it compare to racemic—aka old-fashioned—ketamine?9
Well, this isn’t an article on Ketamine, but it’s not better—it is probably worse:
24 trials representing 1877 participants were pooled. Racemic ketamine relative to esketamine demonstrated greater overall response (RR = 3.01 vs. RR = 1.38) and remission rates (RR = 3.70 vs. RR = 1.47), as well as lower dropouts (RR = 0.76 vs. RR = 1.37).
That is likely to require another article. There is only so much you can read from me in a day!
In summary:
Esketamine is effective for depression that either has not responded to prior treatment OR for someone who has active suicidal ideation at the time they are starting treatment.
It’s not yet approved as a monotherapy—treatment on its own. There are ethical reasons drug trials are done in this sequential manner.
It requires 2 hours per visit for administration, which can only be done in a physicians office with specific certification.
Thanks for reading! Please subscribe and share with your friends and colleagues if you find it helpful.
Prior drug-themed articles include:
Effexor, Buspar, Risperdal, Zyprexa, Neurontin, Xanax, Klonopin, Paxil, Prozac, Clozaril, Lamictal, Lithium, Latuda, Ambien, and generally Benzos, specifically maybe Benzos leading to death by suicide, Geodon, Zoloft, Auvelity, Abilify I, Abilify II: Mania; Abilify III: Bipolar Depression, Akathisia (aka Abilify IV), Abilify V: Federal Enforcement, Abilify VI
I know —a little overboard on Abilify. Just like a psychiatrist!
Evans, V. D., Arenas, A., Shinozuka, K., Tabaac, B. J., Beutler, B. D., Cherian, K., ... & Muir, O. S. (2024). Psychedelic Therapy: A Primer for Primary Care Clinicians—Ketamine. American Journal of Therapeutics, 31(2), e155-e177.
Solomon, T. M., Hajduk, M., Majernik, M., Jemison, J., Deschamps, A., Scoggins, J., ... & Barrow, R. (2023). Evaluating passive physiological data collection during Spravato treatment. Frontiers in Digital Health, 5, 1281529.
Zhang JC, Yao W, Hashimoto K. Arketamine, a new rapid-acting antidepressant: A historical review and future directions. Neuropharmacology. 2022 Nov 1;218:109219. doi: 10.1016/j.neuropharm.2022.109219. Epub 2022 Aug 14. PMID: 35977629.
Papakostas GI, Salloum NC, Hock RS, Jha MK, Murrough JW, Mathew SJ, Iosifescu DV, Fava M. Efficacy of Esketamine Augmentation in Major Depressive Disorder: A Meta-Analysis. J Clin Psychiatry. 2020 May 26;81(4):19r12889. doi: 10.4088/JCP.19r12889. PMID: 32459407.
Reif A, Bitter I, Buyze J, Cebulla K, Frey R, Fu DJ, Ito T, Kambarov Y, Llorca PM, Oliveira-Maia AJ, Messer T, Mulhern-Haughey S, Rive B, von Holt C, Young AH, Godinov Y; ESCAPE-TRD Investigators. Esketamine Nasal Spray versus Quetiapine for Treatment-Resistant Depression. N Engl J Med. 2023 Oct 5;389(14):1298-1309. doi: 10.1056/NEJMoa2304145. PMID: 37792613.
Fu DJ, Ionescu DF, Li X, Lane R, Lim P, Sanacora G, Hough D, Manji H, Drevets WC, Canuso CM. Esketamine Nasal Spray for Rapid Reduction of Major Depressive Disorder Symptoms in Patients Who Have Active Suicidal Ideation With Intent: Double-Blind, Randomized Study (ASPIRE I). J Clin Psychiatry. 2020 May 12;81(3):19m13191. doi: 10.4088/JCP.19m13191. PMID: 32412700.
Ionescu DF, Fu DJ, Qiu X, Lane R, Lim P, Kasper S, Hough D, Drevets WC, Manji H, Canuso CM. Esketamine Nasal Spray for Rapid Reduction of Depressive Symptoms in Patients With Major Depressive Disorder Who Have Active Suicide Ideation With Intent: Results of a Phase 3, Double-Blind, Randomized Study (ASPIRE II). Int J Neuropsychopharmacol. 2021 Jan 20;24(1):22-31. doi: 10.1093/ijnp/pyaa068. PMID: 32861217; PMCID: PMC7816667.
Ross, E. L., & Soeteman, D. I. (2020). Cost-effectiveness of esketamine nasal spray for patients with treatment-resistant depression in the United States. Psychiatric Services, 71(10), 988-997.
Bahji A, Vazquez GH, Zarate CA Jr. Comparative efficacy of racemic ketamine and esketamine for depression: A systematic review and meta-analysis. J Affect Disord. 2021 Jan 1;278:542-555. doi: 10.1016/j.jad.2020.09.071. Epub 2020 Sep 23. Erratum in: J Affect Disord. 2020 Nov 20;: PMID: 33022440; PMCID: PMC7704936.


Thank you for another good read
read
Is there a book you would recommend if one was to learn about organic chemistry applied to medecine ?
Thank you