Welcome to The Frontier Psychiatrists. It’s a newsletter. This is another installment in my series of articles on medications and their history—a sassy prescribing guide from someone… who is moving on to neuromodulation as fast as humanly possible.
Venlafaxine, brand name “Effexor,” was approved by the FDA in 1993, and the extended-release version, Effexor XR, was approved in 1997.
INDICATIONS AND USAGE
Effexor (venlafaxine hydrochloride) is indicated for the treatment of major depressive disorder.
The efficacy of Effexor in the treatment of major depressive disorder was established in 6-week controlled trials of adult outpatients whose diagnoses corresponded most closely to the DSM III or DSM-III-R category of major depression and in a 4-week controlled trial of inpatients meeting diagnostic criteria for major depression with melancholia (see CLINICAL TRIALS).
The molecule looks like this:
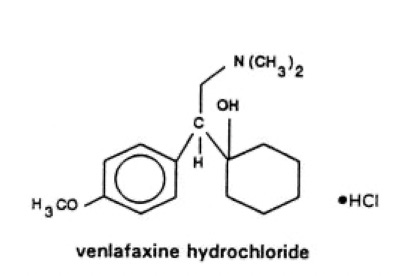
Effexor is metabolized in the liver into its active metabolite, desvenlafaxine. You can now also be prescribed this as Pristiq. Without too much of a digression, hepatic (aka Liver) metabolism matters because, especially for a drug like Effexor, which is made more active by this metabolic process, the variability in the population of humans in how they process any drug matters. The liver has a series of proteins called enzymes, and they are called the cytochrome p450 family. They convert a drug into another molecule. That next molecule is either more or less active in the body than the parent compound. Effexor is processed at the 2D6 enzyme, which has a wide range of variability in humans. There are papers on the 2d6 genetics you have1 predicting if this medicine will be helpful to you.
Some people process it slowly. Some people clear it out fast. You can't know in advance without a genetic test that didn’t exist in 1993 or 1997—and is still not in common use.


