Oh god. Not another SSRI. I promise you, there is something unexpected in this article. YES. Yes, precious, another one. Lexapro is the brand name for escitalopram, which is the “left-handed” enantiomer of the drug citalopram, a.k.a., Celexa. The FDA approved it on August 14, 2002. This is the classic “me too” antidepressant. It garnered the following indications subsequently:
Acute and Maintenance Treatment of Major Depressive Disorder (MDD) in adults and adolescents aged 12-17 years
Acute Treatment of Generalized Anxiety Disorder (GAD) in Adults.
What is special about Lexapro? It’s a much…smaller pill. Celexa, the mixture of the left and right-handed molecules of citalopram, is dosed in 10, 20, 30, and 40mg tablets. Lexapro? 5, 10, 20mg. It’s half the milligrams! That sounds like less medicine, which might not be important to you, but in a drug for anxious people, saying it’s as little as 5mg? That sounds less. Do you want to take 80 milligrams of Prozac? How about 200 mg of Zoloft? No? Sounds like too much. I know…what would you say to just 5 mg of Lexapro?
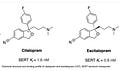
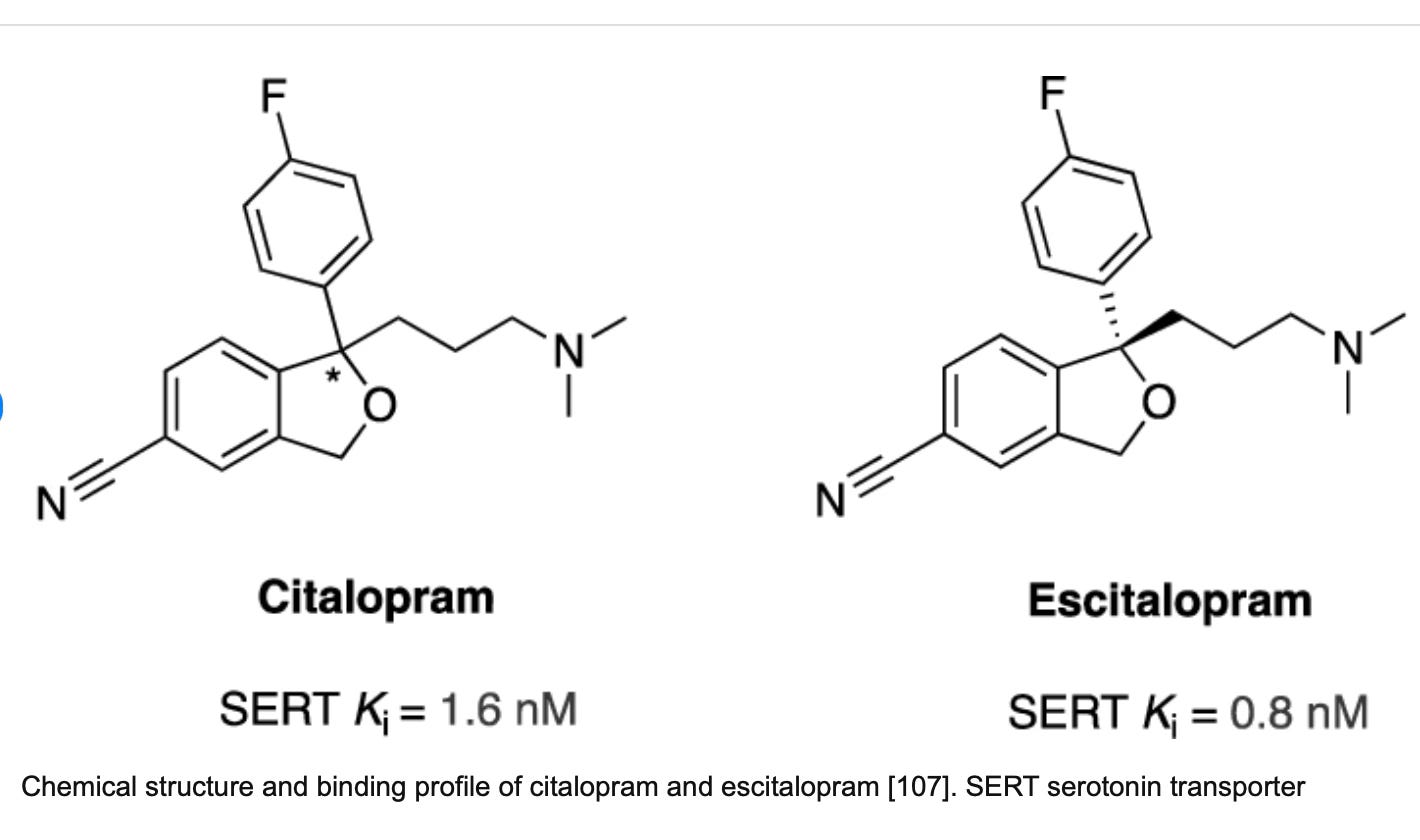
Exactly. It’s huge, in that it is so small. The escitalopram molecule has a stronger binding affinity than the citalopram molecule (which is a racemic mixture of left and right-handed molecules):
A brief personal aside—what is my favorite thing, after connecting with my fabulous readers, that comes from authoring this publication? It is how deep the wormhole of the unknown goes. For example, there has been a randomized controlled trial of generic vs. brand name Lexapro. Yes. That happened. Spoiler alert: there was no difference:
In the intention-to-treat analysis (i.e., including all patients), the mean (s.d.) drop in the HAMD total score at the end of the 8th week of treatment was 13.9 (8.2) in the generic escitalopram group and 14.3 (8.1) in the Lexapro group (t=0.44, p=0.664). The proportions of patients responsive to treatment (i.e., >50% drop in total HAM-D score) were 69% and 67% in the generic escitalopram group and Lexapro group, respectively…1
What is more shocking is the following:
There were 35 (27%) dropouts during the 8 weeks of treatment in the generic escitalopram group and 32 (25%) in the Lexapro group.
Not only were the dropout rates high, but there were suicide events—including attempts in this clinical trial. This occurred in both groups and, most tragically, two deaths by suicide occurred—a rare event in clinical trials. Suicidal individuals are routinely screened out at enrollment, and yet, in this trial:
The occurrence of 4 suicidal events (2 deaths and 2 attempts) in the 260 patients (1.5%) treated with escitalopram during the first 35 days of treatment may be a statistical outlier, but the rates of suicidal events should be re-assessed in larger studies with longer follow-up times.
It’s worth mentioning this was in part a pharma-funded trial conducted in China, but by the generic manufacturer:
Funding: Funding for this study was received from three sources: (a) the National Science and Technology Major Project for Investigational New Drugs subproject titled ‘Clinical Technological Platform for Evaluation of New Drugs in Psychiatry’ (CPEP number 2012ZX09303-003); (b) the Shanghai Jiao Tong University School of Medicine 985 Project titled ‘Standardized Platform for Clinical Testing of Neuropsychiatric Medications’; and (c) the Jiangsu Nhwa Pharmaceutical Corporation Limited.
This effect has demonstrated itself in other studies, to a more limited degree, such as a placebo-controlled trial of Lexapro vs. placebo in youth:
Occurrence of suicidal behavior and/or suicidal ideation assessed by C-SSRS was 10.9% (14/128) for placebo and 14.5% (19/131) for escitalopram.2
There wasn’t a statistically significant difference…however, the studies are underpowered to determine the rates of rare adverse effects.
There is more on this thread to pull…stay tuned!
Yu Y, Li H, Wang B, Li K, Xu X, Shi J, Gao C, Tan Q. Efficacy and safety of generic escitalopram versus Lexapro in the treatment of major depression: a multicenter double-blinded randomized controlled trial. Shanghai Arch Psychiatry. 2013 Apr;25(2):107-15. doi: 10.3969/j.issn.1002-0829.2013.02.007. PMID: 24991142; PMCID: PMC4054541.
Findling, R. L., Robb, A., & Bose, A. (2013). Escitalopram in the treatment of adolescent depression: a randomized, double-blind, placebo-controlled extension trial. Journal of child and adolescent psychopharmacology, 23(7), 468-480.


Thank you excellent article